Battery
How do you define the stage of civilization? There are many ways, but some scholars say, we can divide it according to how much energy the civilization uses. For our civilization to continue
and expand, we need the advancement of energy generation/harvest/transport/storage technology. Nuclear fusion and power generation technology using solar panels are developing very rapidly. On
the other hand, innovation in transportation/storage technology has been developing slowly since the first lithium-ion battery was developed in the 1990s. In particular, Batteries are a field
of research on the interface between electrodes and electrolytes, so there are many challenges to improving performance.
There are a number of issues that can be an obstacle to the development of secondary batteries. To simplify it, (1) energy density (2) fast charging (3) operability in a wide temperature range
is required for innovation in secondary batteries. The problem is that (4) price (5) safety (6) stability is sacrificed to secure them. In our lab, we test various materials using
electrochemical/physical chemistry principles to re-establish this trade-off relationship.
Battery-‹Ionics›
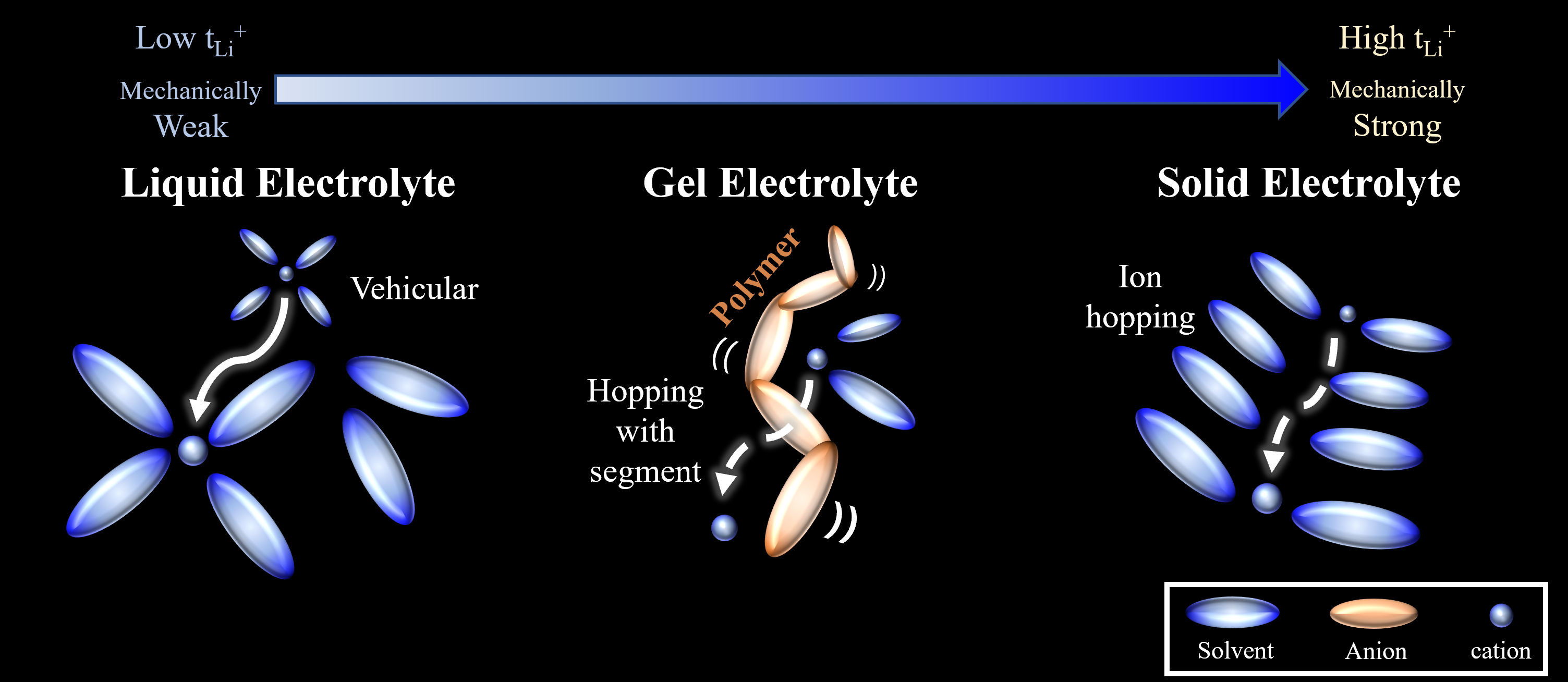
Among the battery components, Ionics considers the electrolyte and electrolyte-electrode interphase for revealing behavior of ions. Our laboratory presents various phase of electrolytes;
general liquid electrolytes, challenging solid electrolytes, and gel electrolytes with intermediate properties.
We are designing liquid electrolytes to secure the stability at various harsh atmosphere. (1) Electrochemically harsh atmosphere, where using lithium metal anodes/ high
voltage cathode for high energy density and (2) Thermal harsh atmosphere, operating at cryogenic temperature (< -20oC) for winter to sub-marine and space. We want to find and solve the
deterioration mechanism of the battery. Reducing energy barriers such as SEI (Solid-Electrolyte Interphase) layer design and charge transfer resistance are our main strategies.
Due to the physical properties of liquid electrolytes, solvents/anions are continuously supplied to the electrodes. In addition, weak mechanical strength does not prevent lithium dendrites
from growing. These require a solid ion conduction layer, such as SEI/CEI, and ultimately a solid electrolyte is proposed. We are improving the problem of conventional solid
electrolytes and also finding new “ionic conductive solid” as single ion conductor for solid electrolyte. Our insight from in-situ solidification can improve the contact resistance problem and
processability. For verifying their ion transporting, electrochemical analysis and structure analysis (crystallography) are used and developed in our group.
Solid electrolytes, on the other hand, have so different physical properties that they can cause various problems such as ionic conductivity and manufacturing process.
Therefore we are developing Gel Polymer Electrolytes (GPEs, polymers+liquid electrolyte) that can be a bridge between liquid electrolytes and solid electrolytes. GPEs guarantee not also the
low interfacial contact resistance but excellent mechanical properties and electrochemical performances such as ion kinetics. Our group reported a GPE characterized by in situ gelation within
battery cells without additional gelating agents. In our goal on GPE is maintaining the fundamental battery performance while utilizing enhancement of additional functionalities such as flame
retardant, cyclability, and energy density.
Battery-‹Electrodics›
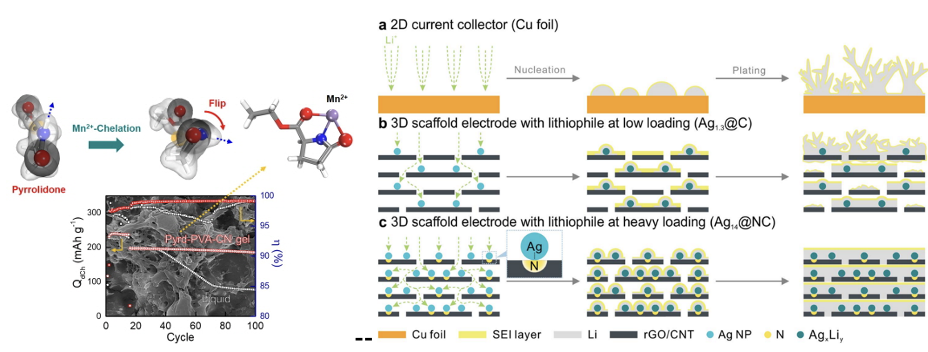
Electrodics is concerned the physical chemistry of electrically charged interface. Electrodes can define the performance of the battery. The energy density of a battery can be increased by
using a positive electrode in a high voltage range, a negative electrode in a low voltage range, or an electrode in a high capacity. The problem with these electrodes is the lack of
stability/safety. These problems are represented by volume expansion, transition metal-ion dissolution and gas evolution in anodes.
To prevent these things, we want to reveal the degradation mechanism of the cell and suppress it. For that purpose, we are working on material improvement based on physical chemistry and
electrochemical. Frameworks or interface layer, based on various materials from polymers to organic and inorganic materials, are formed on the electrode surface to derive (1) physical
strength, (2) lithium behavior control, and (3) side reaction inhibition effects. We want to solve the problem of the next generation of anode/cathode materials by exploring this framework.
Reference paper :
J. Mater. Chem. A, 2018, 6, 22483
J. Mater. Chem. A, 2022, 10, 1768

